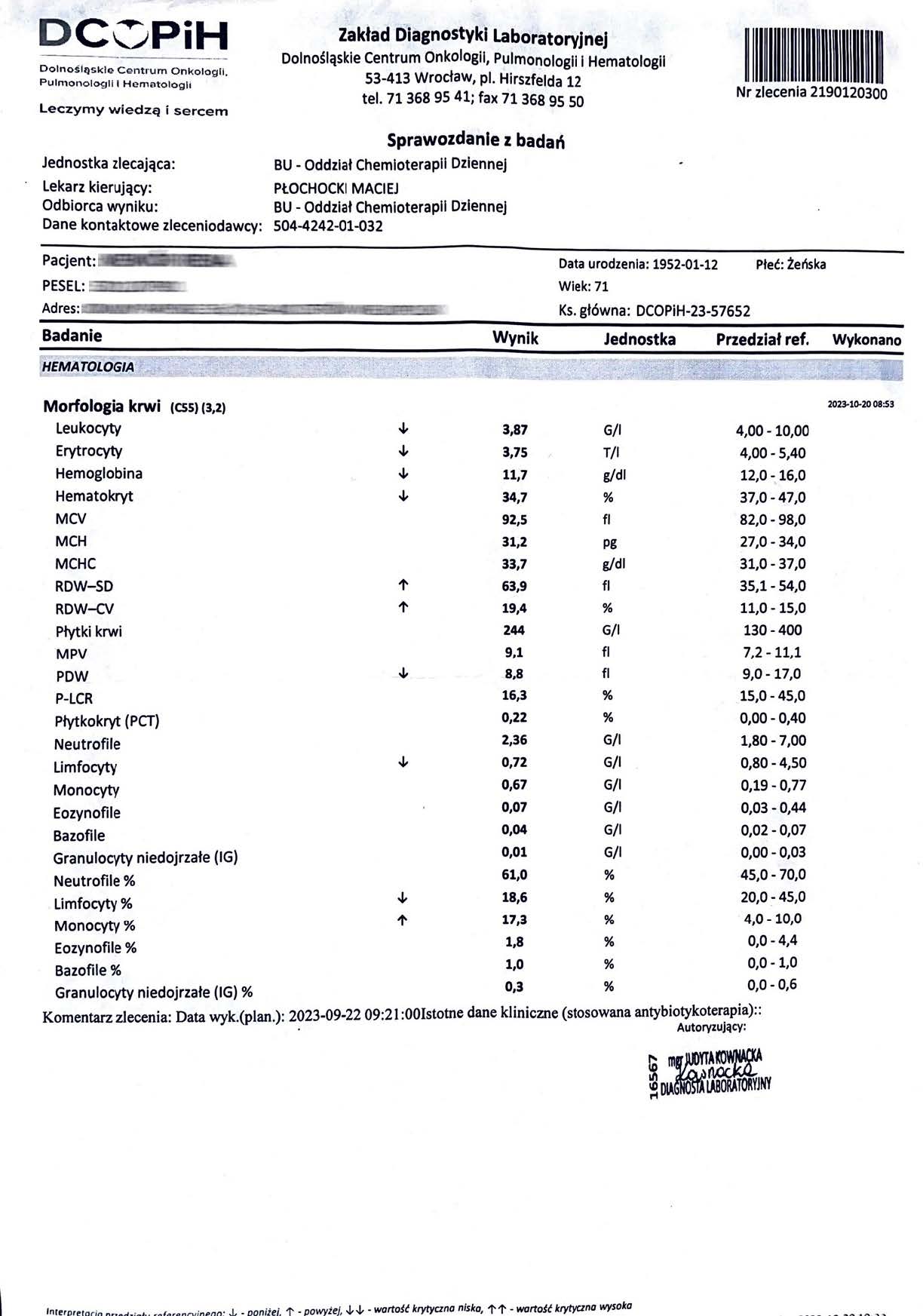
Confirmation of the toxicological safety of the intracellular ATP preparation
The results of ATP measurements in the bloodstream and within granulocytes, obtained during long-term use of the preparation, provide significant indirect evidence of the lack of mitochondrial toxicity and the overall safety of the preparation.
Increase and stabilization of ATP levels in the blood -
ATP levels increased from ~2.8 µM (lower limit of normal) to 7.59 µM (upper limit, value typical for high-energy tissues), and after discontinuation remained above the baseline value (~3.67 µM). The lack of a decrease in ATP – or even a significant and sustained increase – is inconsistent with the profile of any substance toxic to mitochondria (complex I-V inhibitors, rotenone, antimycin, many oncological drugs).
Mitochondrial toxins cause a rapid and progressive decrease in ATP – the observed effect is the opposite.
- Significant increase in ATP in granulocytes (>100%)
Granulocytes (neutrophils) are one of the most sensitive cell populations to mitochondrial toxicity. A decrease in ATP in neutrophils by >30–40% is considered an early marker of cytotoxicity (OECD, ICCVAM tests). The observed increase in ATP in granulocytes
indicates not only a lack of toxicity, but also active support for the bioenergetics of immune cells – which is very rare and constitutes a positive signal of immunological safety.
Compliance with the criteria for non-mitochondrial toxicity -
Toxicological guidelines (FDA, EMA, ICH S8, OECD) consider a substance non-toxic
to mitochondria if it meets at least several of the following characteristics (all met
by the ATP preparation):
* No decrease in mitochondrial membrane potential and ATP under conditions of long-term
exposure → the observed increase in ATP is strong evidence.
* No increased mtROS (mitochondrial reactive oxygen species) production →
No decrease in ATP and no clinical symptoms indicate stabilization of the respiratory chain.
* Lack of activation of apoptotic mitochondrial pathways (cytochrome C, caspase-9)
→ no decrease in ATP and no adverse effects are indirect confirmation.
* Maintaining or improving the function of cells with high energy demand
(granulocytes, erythrocytes, myocytes) → an increase in ATP in granulocytes meets this condition to an outstanding degree.
Conclusion:
Long-term use of the ATP preparation not only does not cause a decrease in mitochondrial ATP (as would be typical for mitochondrial toxins), but leads to its significant and sustained increase – both in the blood and in granulocytes. This profile is
contrary to any known mechanism of mitochondrial toxicity and meets
the criteria for considering the substance safe for mitochondria under conditions of long-term
exposure. These results may constitute an important element of the preclinical and clinical data package confirming the safety of the preparation.
EFFECT OF THE PREPARATION ON HYPOECHOGENIC TUMORS
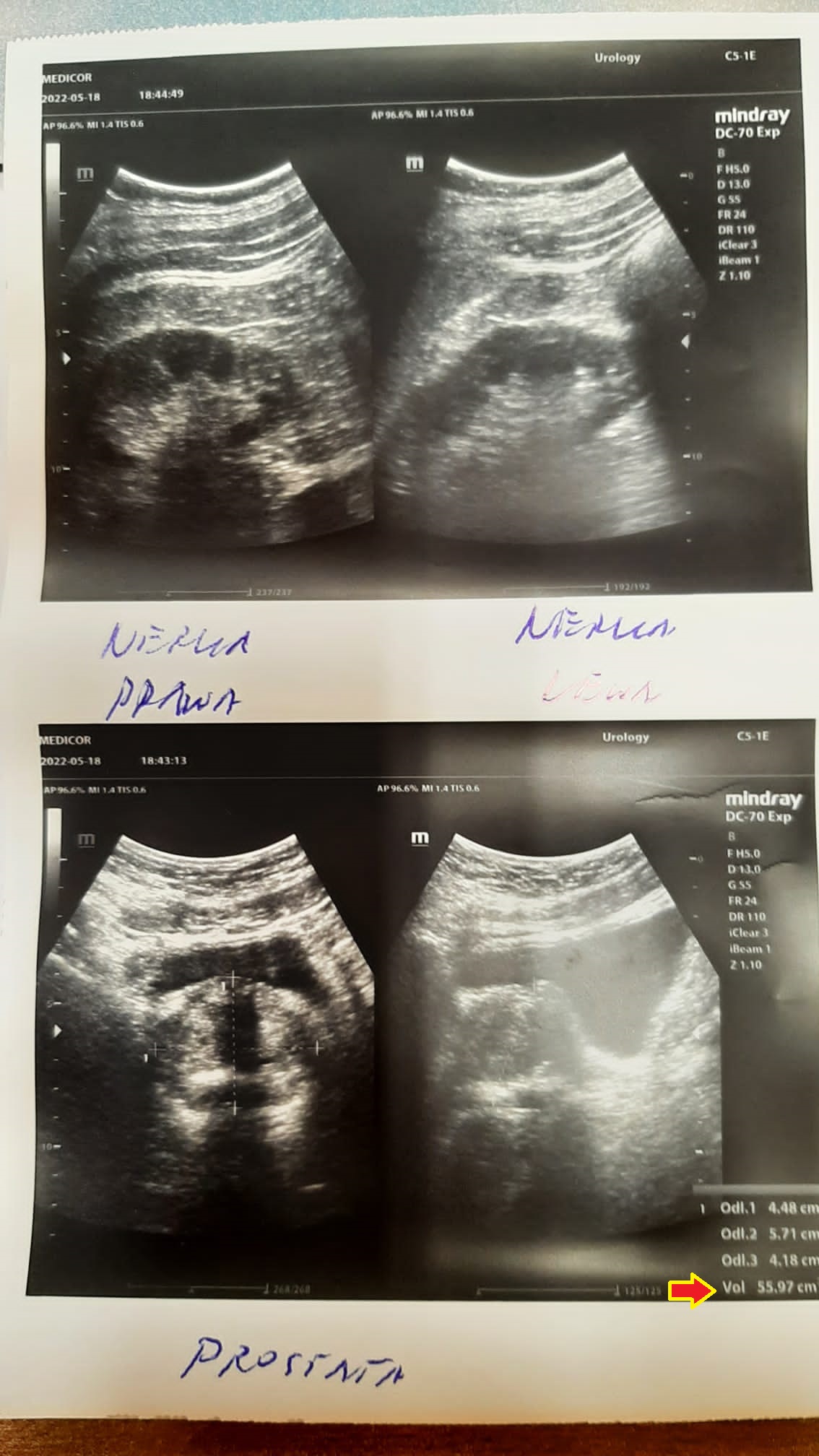
A 66-year-old man with a hypoechoic tumor measuring initially 16x10 mm and a prostate measuring 55x47 mm began treatment with the product. After three months, the first changes were observed: the tumor had shrunk to 6x7 mm and the prostate itself had decreased in size to 50x40 mm.
An examination after six months of treatment revealed no tumor, confirmed by imaging, and the prostate had reduced to the optimal size (48x61 mm), typical for this age group. The complete disappearance of the tumor in such a short time without the intervention of chemotherapy or radiotherapy indicates deep cellular regeneration. By stimulating mitochondria, the product delivers an increased amount of mitochondrial ATP, which leads to apoptosis of damaged prostate cells. The cellular regeneration process, by stimulating and protecting the structure and function of mitochondria, reverses uncontrolled cell division, indicating anti-cancer effects without toxicity.
EFFECT OF THE PREPARATION IN THE CASE OF THYROID NODULES
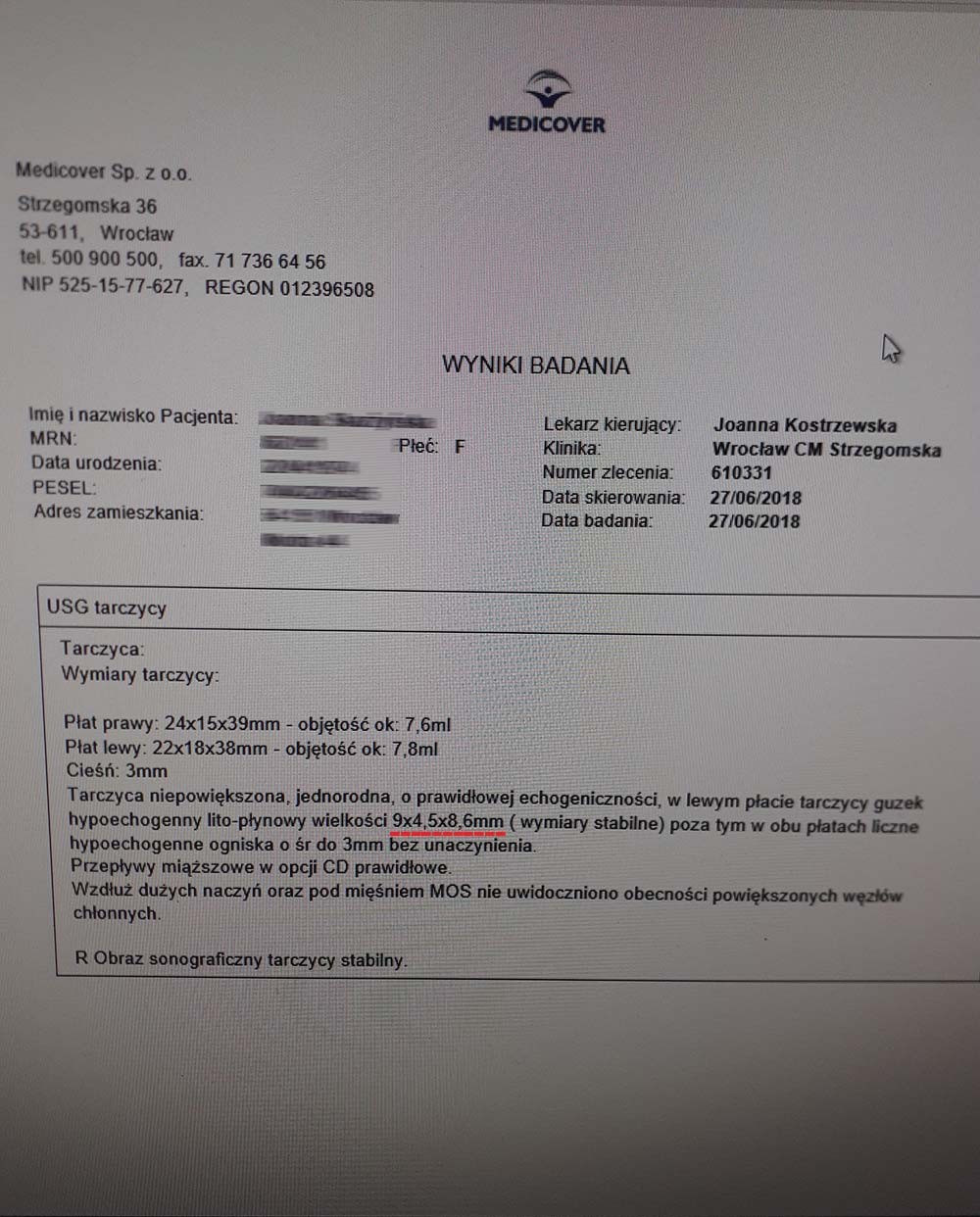
A 46-year-old woman was diagnosed with thyroid nodules measuring 9x4 mm and 5x8 mm before receiving the product. After just one month of treatment without additional medication, the nodules decreased to 7x4 mm and 5x3 mm. The rapid reduction of nodules (~20-65%) without surgical or pharmacological intervention indicates cellular repair at the mitochondrial level, which prevents abnormal cell division and tumor development. The product increases mitochondrial efficiency in ATP production, stimulating apoptosis of damaged cells while simultaneously promoting regeneration and the formation of healthy structures. This is particularly valuable in the case of benign tumors, where the standard procedure is merely to observe changes, which often leads to the development of much more difficult-to-manage abnormalities.
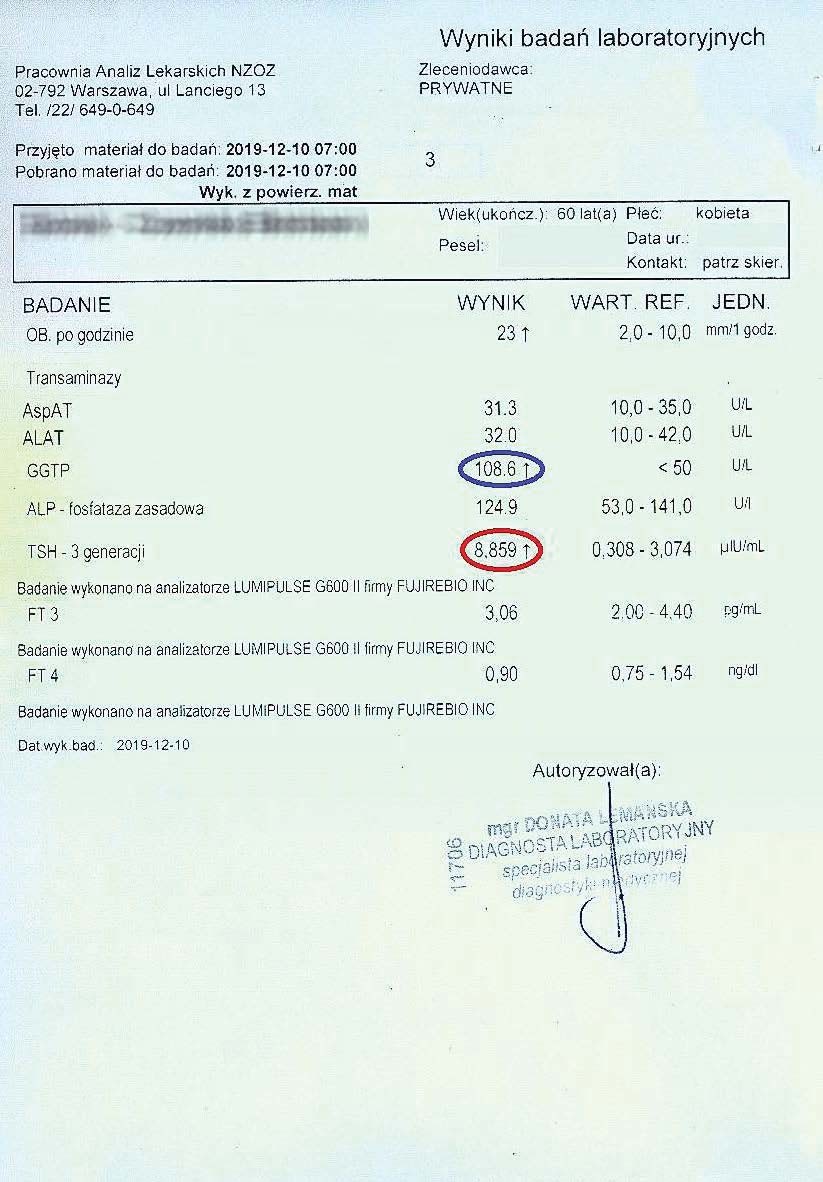
EXAMPLE OF THE EFFECT OF THE PREPARATION ON HYPOTHYROIDISM
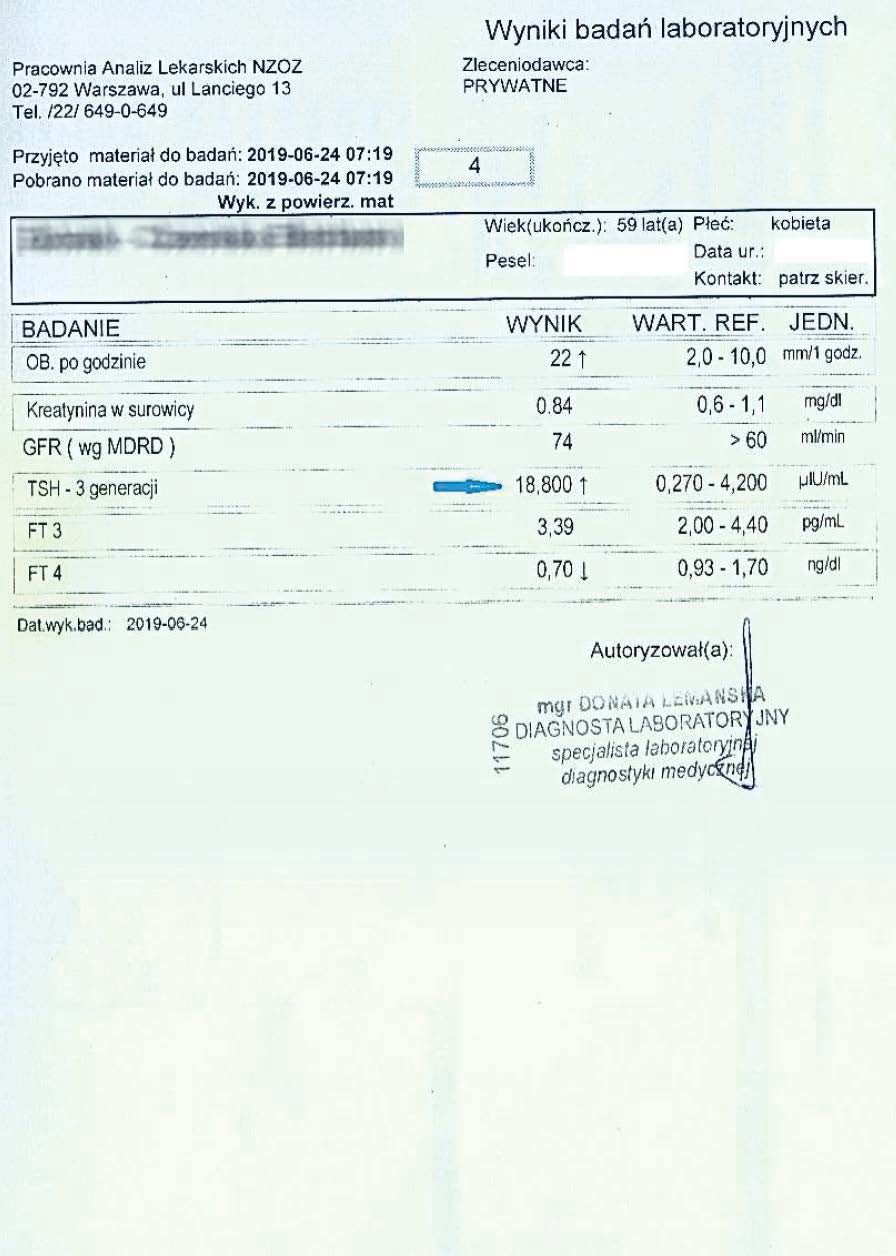
A 59-year-old woman with hypothyroidism, not taking levothyroxine. She took the preparation for over 3 months. While maintaining the natural cell cycle and constantly stimulating mitochondria, she achieved the following treatment results:
After noticing a significant improvement in her overall well-being, the woman decided to have her liver function tests performed more frequently and have her liver function tests monitored.
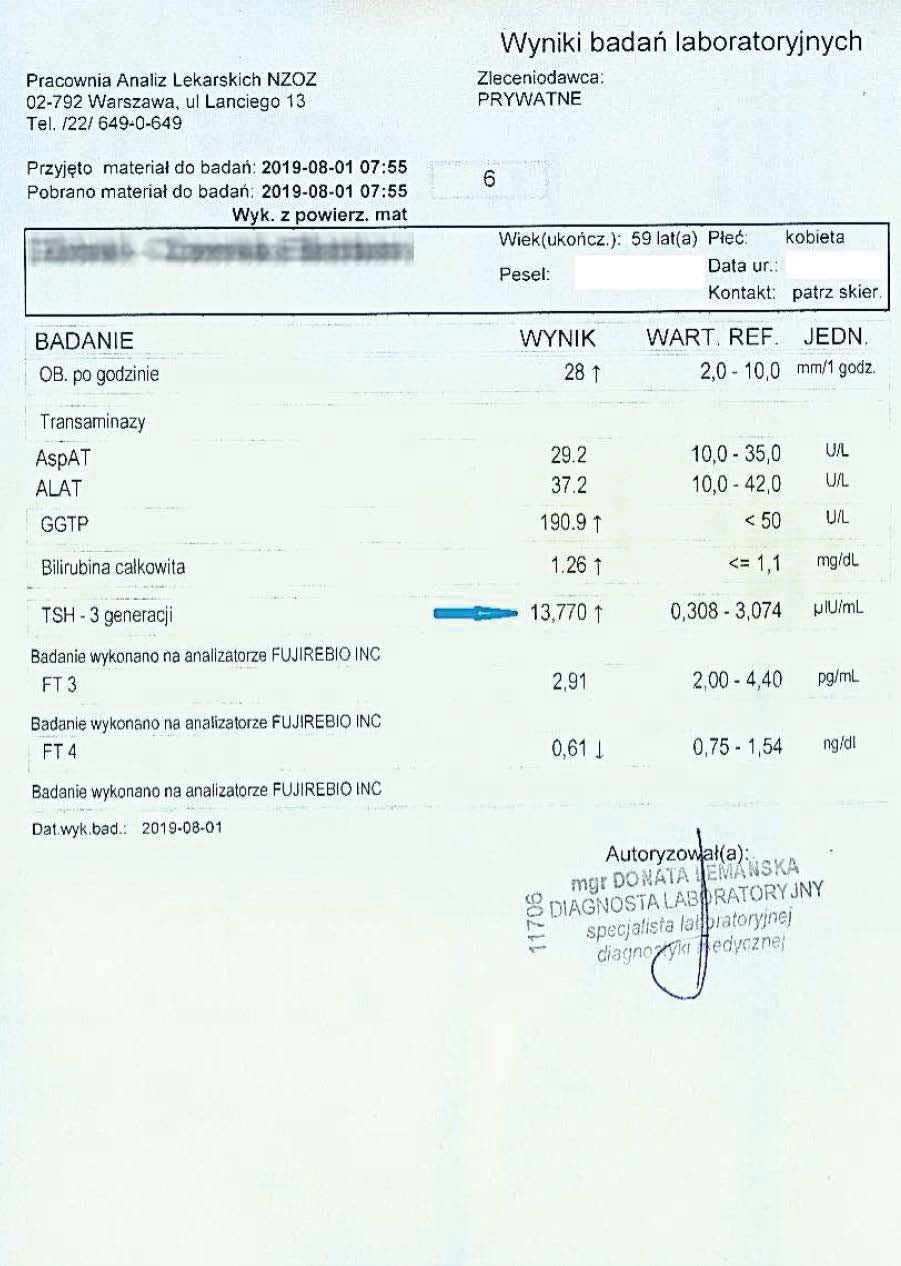
After another two months, the woman had her tests performed again. A decrease in TSH, reduced inflammation, and a decrease in the liver marker GGTP were visible.
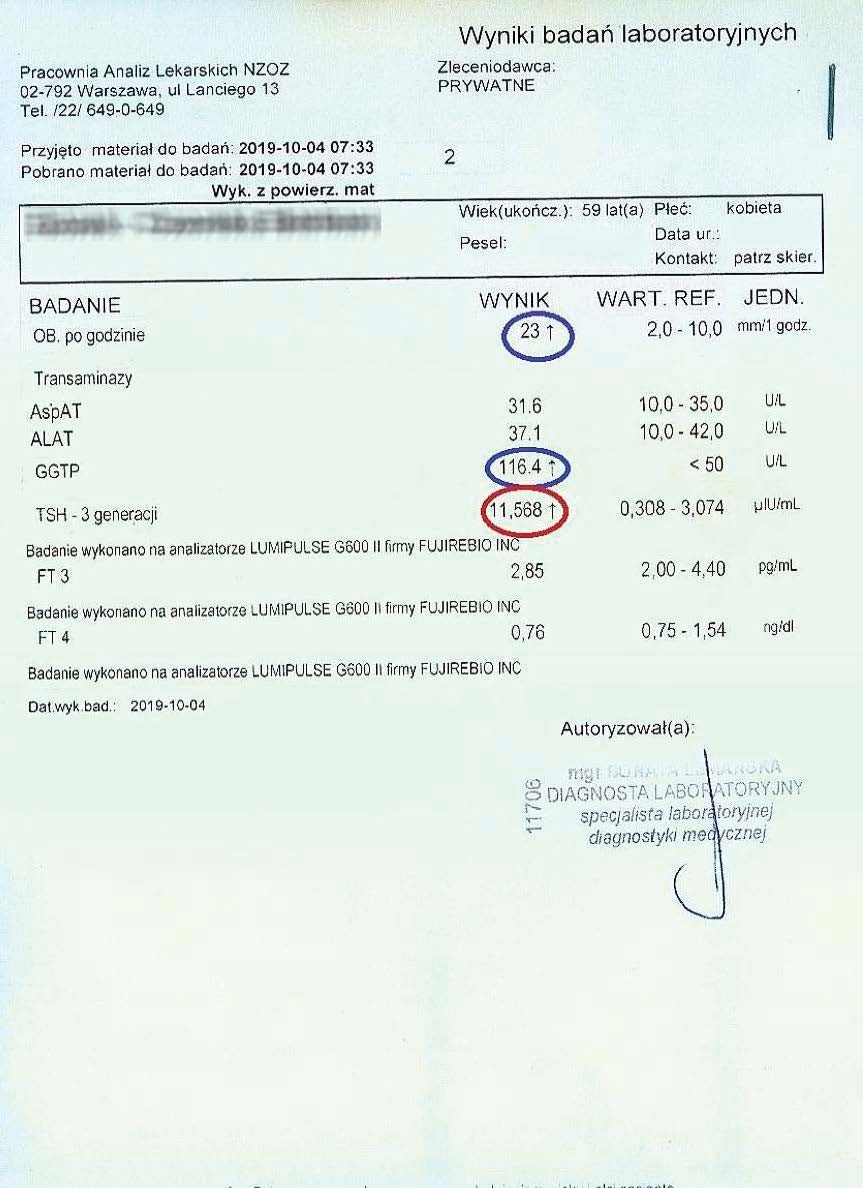
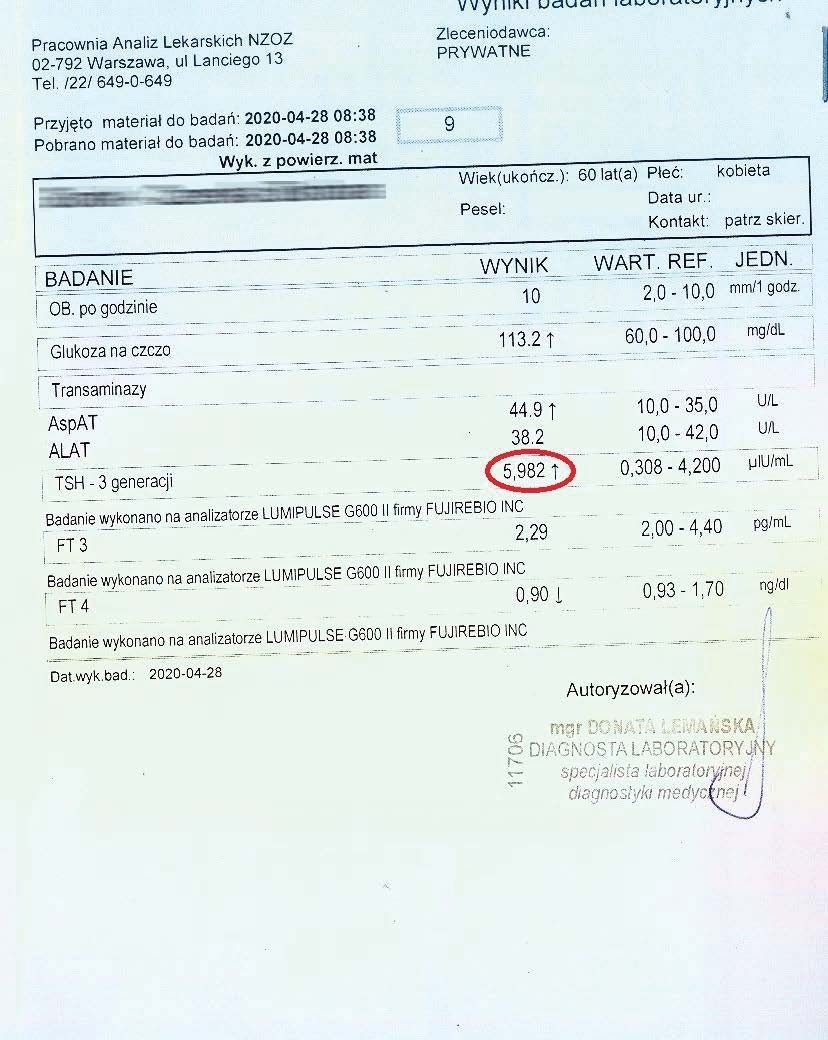
Successively, a steady decline in both liver and thyroid parameters could be observed every two months. Subsequent tests confirmed a further decline in these parameters.
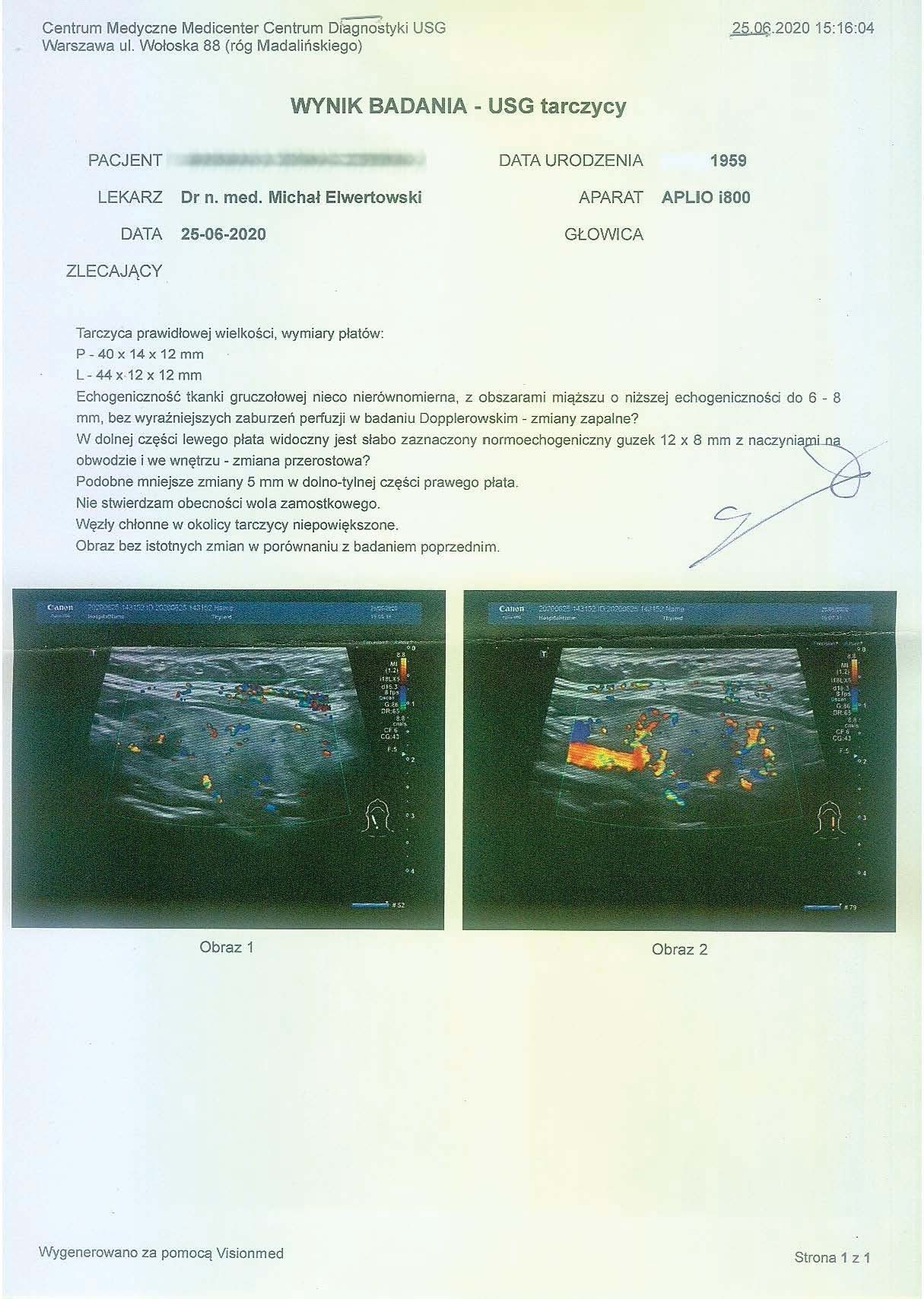
After over a year of treatment, tests showed further progress in reversing the thyroid autoimmunity, also confirmed by an ultrasound of the organ.
By promoting mitochondrial regeneration, the preparation provides an energy base for hormone production and organ regeneration. The lack of pharmacological intervention demonstrates effectiveness even in older people, where regeneration is slower and anabolic reactions are significantly impaired. Systemic effects can also be observed, with a decrease in liver markers, clearly demonstrating improved overall body function.
-
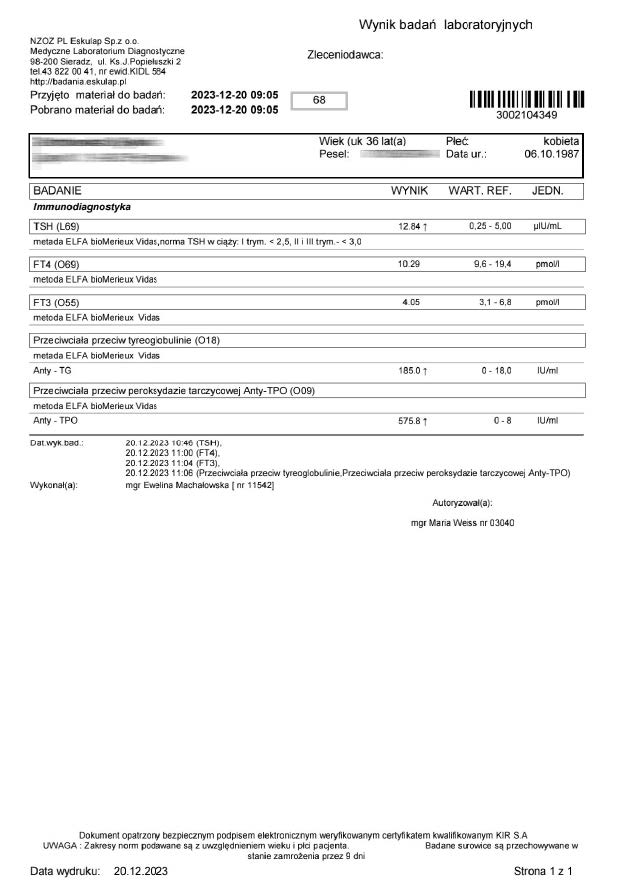
A 36-year-old woman began treatment with the product due to deteriorating well-being and high TSH and anti-TPO levels, clearly indicating hypothyroidism. Levothyroxine was not used. After just 12 days of using the product, a significant decrease in thyroid parameters and antibodies was noted.
The results of reversing autoimmune processes go beyond typical treatment and demonstrate that the product, by strengthening and stimulating mitochondria, works through energetic optimization, enabling long-term regeneration across various age groups.
EFFECT OF THE PREPARATION ON BREAST CANCER
A 71-year-old woman with stage IV breast cancer, advanced metastasis. During treatment, Ca 15-3 markers decreased from 140.0 U/ml to 78.3 U/ml and CEA markers decreased from 11.8 ng/ml to 8.4 ng/ml in just three months. Bone marrow stability was observed. The results indicate inhibition of progression, and additional features were noted that complement pharmacological therapy, making this an example of supportive systemic treatment.
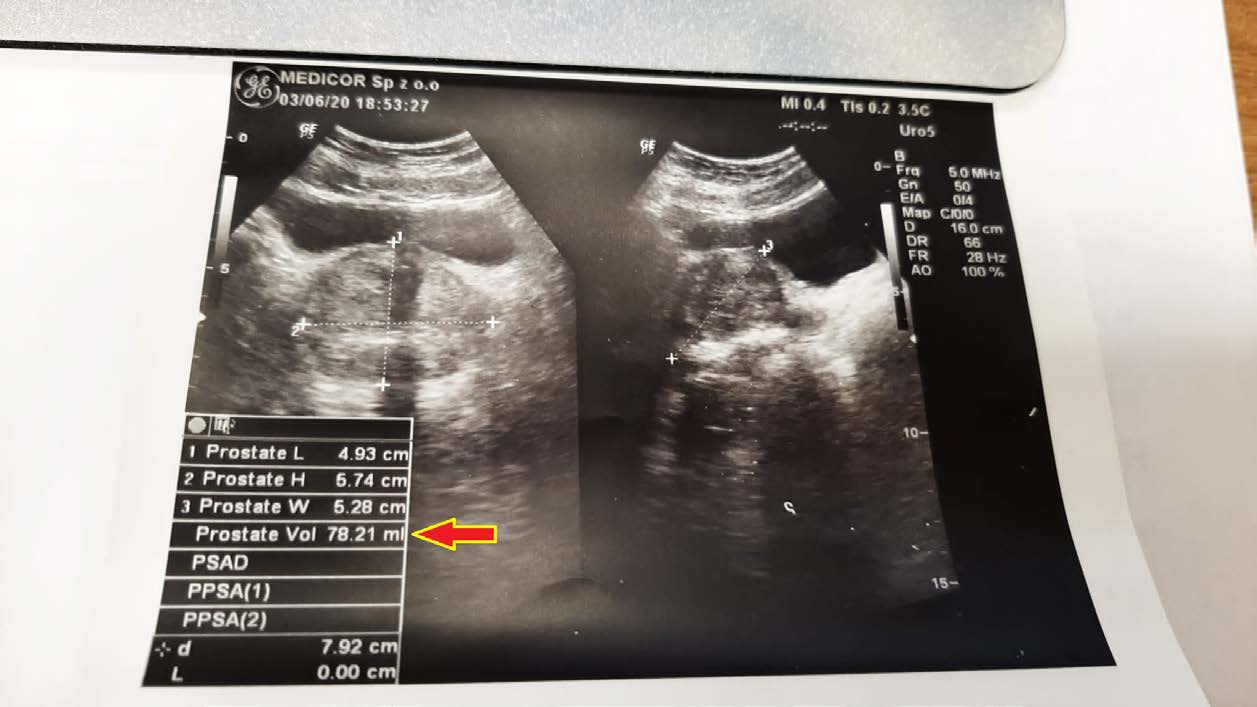
EFFECT OF THE PREPARATION ON PROSTATIC HYPERPLASIA
A 50-year-old man with an enlarged prostate, although mild hyperplasia, experienced pain and discomfort when urinating. His prostate volume at diagnosis was 78.21 ml (78.21 cm3). After unsuccessful attempts with finasteride, an attempt was made to reduce the size of the prostate with a drug. As seen in follow-up ultrasound images, the reduction was significant, reaching 55.97 ml (55.97 cm3) after just 2.5 months of therapy.
EFFECT OF THE PREPARATION ON GOUT
Gout in a 56-year-old man, with a dramatic drop in uric acid. The pre-therapy value was 9.0 mg/dl, accompanied by cyclical gout attacks. However, the history of the disease began much earlier than the first examination. In 2022, therapy with the preparation began, and after 3 months of use, a decrease in uric acid was noted to 7.9 mg/dl. After another 6 months of use, the uric acid level decreased to 3.6 mg/dl. As the uric acid level decreased to the desired level, gout attacks ceased, and the effect is sustained. A 60% reduction in uric acid demonstrates a reversal of urate accumulation. The preparation increases mitochondrial ATP in kidney and liver cells, improving purine metabolism and filtration, which removes urate crystals and reverses arthritis. Standard treatment with allopurinol and colchicine requires lifelong treatment with side effects that affect daily functioning, while in the case of this preparation there are no side effects and a relatively quick response of the body.
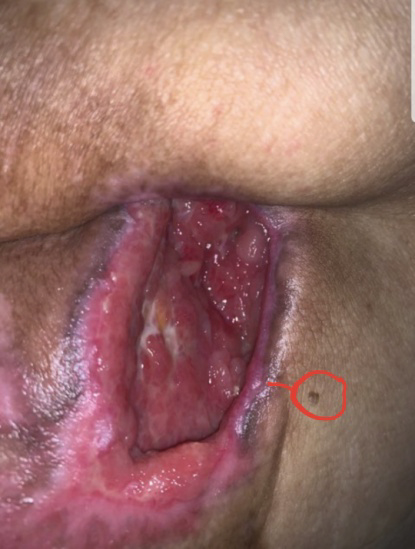
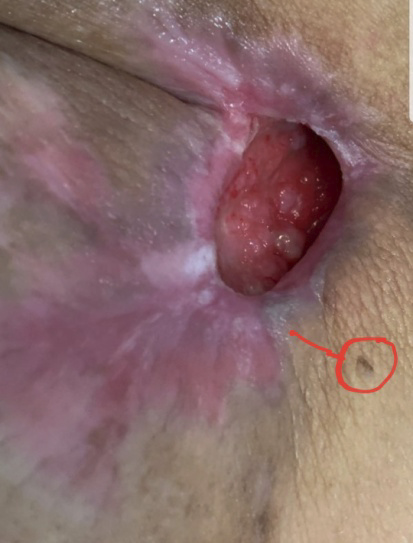
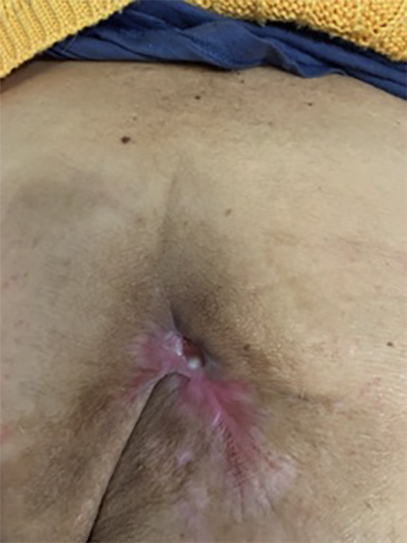
EXAMPLE OF CHRONIC WOUND HEALING
A chronic wound developed as a result of the removal of a cancerous tumor two years after surgery. Its diameter was 5 cm. The next two photos are taken two and three months after treatment. After three months of treatment, the wound had completely healed. The red dot is the reference point.
Scientific Publications
ATP in the Treatment of Advanced Cancer
Edward H. Abraham, Anna Y. Salikhova, and Eliezer Rapaport
Dartmouth Medical School, Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire 03756
https://www.researchgate.net/publication/251464267_ATP_in_the_Treatment_of_Advanced_Cancer
Enhancing skin wound healing by direct delivery of intracellular adenosine triphosphate
Benjamin Chiang, M.D.a, Eric Essick, M.S.b, William Ehringer, Ph.D.b, Sidney Murphree, M.D.c, Mary Anne Hauck, M.S.a, Ming Li, B.S.a, Sufan Chien, M.D.a,
Department of Surgery, University of Louisville, Louisville, KY 40202, USA
Department of Physiology, University of Louisville, Louisville, KY 40202, USA
Department of Pathology, University of Louisville, Louisville, KY 40202, USA
https://pubmed.ncbi.nlm.nih.gov/17236849/
Extracellular ATP and Adenosine in Cancer Pathogenesis and Treatment
Anna M. Chiarella, Yun K. Ryu Gulam A. Manji and Anil K. Rustgi
https://pubmed.ncbi.nlm.nih.gov/34074623/
Extracellular ATP Attenuates the Growth of Hormone Refractory Prostate Cancer In Vivo
M Shabbir, C.S. Thompson Burnstock, D.P. Mikhailidis, R.J. Morgan & G. Burnstock
Departments of Urology”‘, Chemical Biochemistry’*’ & The Autonomic Neuroscience Institute’3’,
Royal Free & University College Medical School (Royal Free Campus), London
https://pubmed.ncbi.nlm.nih.gov/18325054/
Health and ergogenic potential of oral adenosine-5′-triphosphate (ATP) supplementation
Ralf J¨ager, Martin Purpura, John A. Rathmacher, John C. Fuller Jr., Lisa M. Pitchford, Fabricio E. Rossi , Chad M. Kerksick
Increnovo LLC, 2138 E Lafayette Pl, Milwaukee, WI 53202, USA
MTI BioTech, Inc., 2711 S. Loop Dr., Suite 4400, Ames, IA 50010, USA
Dept. of Animal Science, Iowa State University, Ames, IA 50011, USA
Metabolic Technologies, LLC, 135 W Main St, Suite B, Missoula, MT 59802, USA
Dept. of Kinesiology, Iowa State University, Ames, IA 50010, USA
Immunometabolism of Skeletal Muscle and Exercise Research Group, Department of Physical Education, Federal University of Piauí (UFPI), 64049-550 Teresina, Piauí, Brazil
Exercise and Performance Nutrition Laboratory, School of Health Sciences, Lindenwood University, St. Charles, MO, USA
https://www.sciencedirect.com/science/article/pii/S1756464621000062
Intracellular delivery of adenosine triphosphate enhanced healing process in full-thickness skin wounds in diabetic rabbits
Jianpu Wang, M.D., Ph.D, Rong Wan, M.D., Ph.D., Yiqun Mo, M.D., Ph.D.,
Ming Li, B.S., Qunwei Zhang, M.D., Ph.D., Sufan Chien, M.D.
Department of Surgery, School of Medicine, University of Louisville;
Department of Environment Health and
Occupational Health Sciences, School of Public Health and Information Sciences, University of Louisville, Louisville, KY,
USA
https://pubmed.ncbi.nlm.nih.gov/20609726/